United States Molecular Diagnostics Market Report: Comprehensive Analysis and Forecast (2025-2033)
1. Market Overview
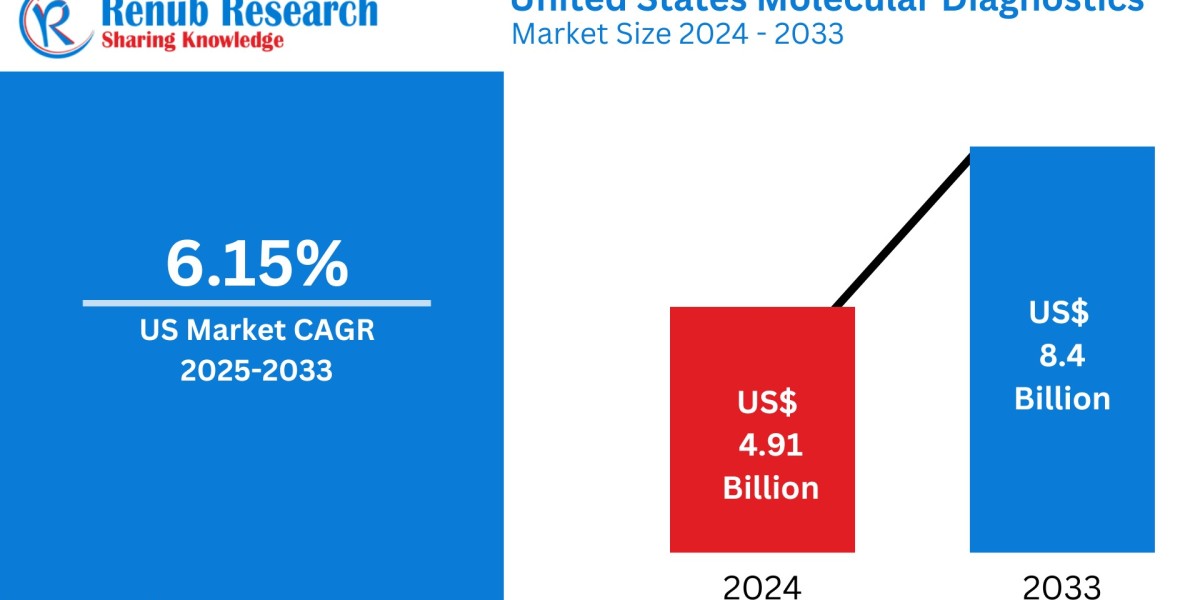
The United States molecular diagnostics market was valued at USD 4.91 billion in 2024 and is projected to reach USD 8.40 billion by 2033, growing at a CAGR of 6.15% from 2025 to 2033. This growth is driven by advancements in diagnostic technologies, the rising demand for personalized medicine, and the increasing prevalence of infectious diseases and cancer. The U.S. remains a global leader in the molecular diagnostics market due to its robust healthcare infrastructure and extensive research and development investments.
2. Market Segmentation
2.1 Technology Segmentation
The market is segmented by technology, including:
- Chips and Microarrays
- In Situ Hybridization
- Sequencing
- Mass Spectrometry (MS)
- Polymerase Chain Reaction (PCR)
- Other Technologies
PCR remains the most widely used technology, especially in detecting infectious diseases and genetic disorders. The advent of digital PCR and AI-integrated diagnostic tools is further enhancing accuracy and efficiency.
2.2 Application Segmentation
Molecular diagnostics are applied in various fields, including:
- Pharmacogenomics: Personalized medicine development
- Infectious Disease: COVID-19, HIV, influenza, hepatitis, etc.
- Genetic Disease Screening: Early detection of inherited conditions
- Oncology: Cancer biomarker detection and liquid biopsy applications
- Blood Screening & Microbiology: Ensuring transfusion safety
- Human Leukocyte Antigen Typing: Transplant compatibility testing
3. Market Growth Drivers
3.1 Rising Incidence of Infectious Diseases and Cancer
- The increasing number of cancer cases and the emergence of infectious diseases continue to drive demand for molecular diagnostics.
- The U.S. is expected to have 2,041,910 new cancer cases and 618,120 cancer-related deaths in 2025.
- Advances in next-generation sequencing (NGS) and liquid biopsy are aiding early cancer detection.
3.2 Technological Innovation and Automation
- Introduction of CRISPR-based diagnostics and AI-driven molecular testing.
- Increasing investment in automated diagnostic systems to enhance accuracy and reduce human error.
- In January 2024, 3EO Health launched an affordable COVID-19 diagnostic test in the U.S.
3.3 Growth of Personalized Medicine
- The U.S. FDA approved 16 new personalized medicines in 2023, compared to six in 2022.
- Personalized medicine reduces side effects and improves treatment efficacy by targeting patient-specific genetic profiles.
Related Report
United States Dental Equipment Market
Europe In-Vitro Diagnostics Market
4. Market Challenges
4.1 High Costs and Reimbursement Issues
- Expensive equipment and reagents make molecular diagnostic tests costly.
- Insurance coverage remains inconsistent, limiting patient access to advanced diagnostics.
4.2 Regulatory Hurdles
- Stringent FDA approval processes delay market entry.
- Laboratory-developed tests (LDTs) face regulatory uncertainties, affecting their adoption.
5. Key Market Segments
5.1 PCR Molecular Diagnostics Market
- PCR remains the gold standard in molecular diagnostics.
- Digital PCR and multiplex PCR are improving test sensitivity and efficiency.
5.2 Infectious Disease Molecular Diagnostics Market
- High demand for quick pathogen detection.
- Governmental funding in pandemic preparedness and antimicrobial resistance research boosts market growth.
5.3 Oncology Molecular Diagnostics Market
- Liquid biopsies and tumor profiling improve early-stage cancer detection.
- The adoption of companion diagnostics supports precision medicine initiatives.
5.4 Instruments Market
- Rising demand for automated and high-throughput diagnostic equipment.
- Increased adoption of portable molecular diagnostic instruments in hospitals and research facilities.
6. Investment & Recent Developments
- Jan 2025: The DDDI FY2022 investment was $263 million for advanced product development and regulatory clearance.
- Jan 2024: ELITechGroup released the GI Bacterial PLUS ELITe MGB® Kit for diagnosing foodborne and hospital-acquired gastrointestinal illnesses.
- Aug 2023: QIAGEN secured U.S. FDA clearance for the Therascreen PDGFRA RGQ PCR kit for gastrointestinal stromal tumor detection.
- March 2023: Lucira Health launched the first at-home COVID-19 and flu diagnostic test in the U.S.
7. Competitive Landscape
7.1 Key Players in the U.S. Molecular Diagnostics Market
- F Hoffmann-la Roche Ltd
- Illumina Ltd
- Hologic Corporation
- Agilent Technologies Inc.
- Qiagen NV
- Myriad Genetics
- Abbott Laboratories
- Danaher Corporation
- Becton, Dickinson and Company
8. Market Forecast and Future Trends
- The U.S. molecular diagnostics market is projected to witness rapid advancements in AI-driven diagnostics, automation, and personalized medicine.
- Investments in point-of-care (POC) testing and telehealth-integrated diagnostic solutions will continue to rise.
- Increasing focus on liquid biopsies and gene editing technologies will revolutionize early disease detection and treatment.
9. Key Questions Answered in This Report:
- What was the U.S. molecular diagnostics market size in 2024?
- What is the forecasted market value by 2033?
- Which segment will experience the highest growth?
- What are the primary challenges affecting the market?
- How is personalized medicine driving demand?
- Who are the major players in the industry?
10. Conclusion
The United States molecular diagnostics market is witnessing substantial growth due to technological advancements, increasing disease prevalence, and a shift toward precision medicine. However, regulatory challenges and cost constraints remain significant hurdles. The future of molecular diagnostics in the U.S. will be shaped by continued innovation, automation, and increasing adoption in personalized healthcare.
For further details or to request a customized report, contact Renub Research at info@renub.com or call +1-678-302-0700 (USA) / +91-120-421-9822 (India).