Global In-Vitro Diagnostics Market Report
Overview
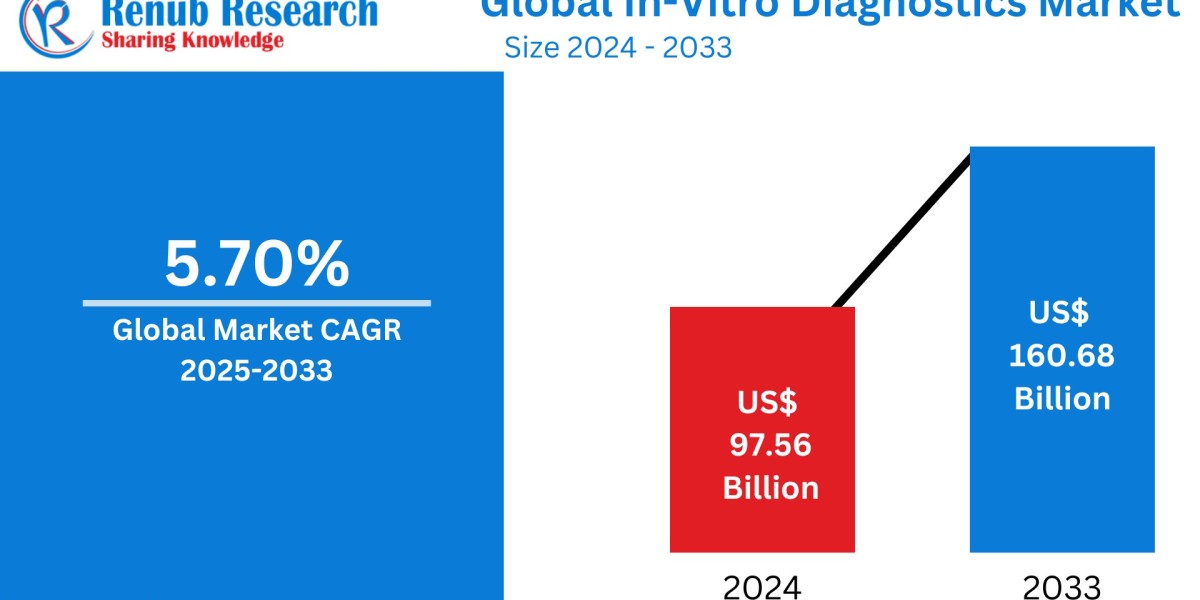
The global in-vitro diagnostics (IVD) market is projected to grow from US$ 97.56 billion in 2024 to US$ 160.68 billion by 2033, registering a CAGR of 5.70%. Key drivers of this market include the rising elderly population, increasing prevalence of infectious diseases, advancements in diagnostic technologies, and a shift toward personalized medicine.
Market Segmentation
By Test Type
- Clinical Chemistry
- Molecular Diagnostics
- Immunodiagnostics
- Hematology
- Others
By Product
- Reagents and Kits
- Instruments
- Others
By Usability
- Disposable IVD Devices
- Reusable IVD Devices
By Application
- Infectious Disease
- Diabetes
- Cancer/Oncology
- Cardiology
- Autoimmune Disease
- Nephrology
- Others
By End User
- Clinical Laboratories
- Point-of-Care Testing Centers
- Academic Institutes
- Patients
- Others
By Region
- North America (United States, Canada)
- Europe (France, Germany, Italy, Spain, United Kingdom, Belgium, Netherlands, Turkey)
- Asia Pacific (China, Japan, India, Australia, South Korea, Thailand, Malaysia, Indonesia, New Zealand)
- Latin America (Brazil, Mexico, Argentina)
- Middle East & Africa (South Africa, Saudi Arabia, UAE)
Growth Drivers
Rising Chronic Illnesses
The prevalence of chronic diseases such as diabetes, cardiovascular diseases, and cancer is increasing globally, driving the demand for in-vitro diagnostic solutions for early detection, monitoring, and management.
Personalized Medicine
The growing focus on personalized medicine is fueling the adoption of IVD tests such as genetic testing and companion diagnostics, enabling tailored treatment approaches based on a patient’s unique characteristics.
Technological Advancements
Innovations in molecular diagnostics, next-generation sequencing, automation, and AI-driven diagnostics are improving the accuracy and efficiency of in-vitro diagnostic tests.
Related Report
United Arab Emirates Red Meat Market
North America Meat Substitutes Market
Challenges
High Competition
The IVD market is highly competitive, with numerous companies offering similar products. Differentiation based on quality and pricing strategies is crucial for market players.
Technological Complexity
The integration of advanced diagnostic solutions with existing healthcare infrastructure remains a challenge, particularly in resource-limited settings.
Regional Analysis
United States
The U.S. IVD market is one of the largest, driven by a high prevalence of chronic diseases, technological advancements, and regulatory approvals from the FDA.
United Kingdom
The UK IVD market is supported by a strong regulatory framework and increasing demand for precision diagnostics, though reimbursement challenges persist.
India
India’s IVD market is expanding rapidly due to rising healthcare awareness, an increasing burden of chronic diseases, and government initiatives for affordable diagnostics.
Saudi Arabia
Saudi Arabia’s market is experiencing significant growth, driven by government healthcare initiatives, an aging population, and increasing medical tourism.
Key Market Players
- Abbott Laboratories
- Agilent Technologies Inc.
- Biomerieux SA
- Bio-Rad Laboratories Inc.
- F. Hoffmann-La Roche Ltd
- Fujifilm Holdings Corporation
- Illumina Inc.
- Qiagen NV
- Quest Diagnostics
- Sysmex Corporation
Report Features
- Base Year: 2024
- Historical Period: 2020 - 2024
- Forecast Period: 2025 - 2033
- Delivery Format: PDF and Excel
- Customization Scope: 20% Free Customization
- Post-Sale Analyst Support: 1 Year (52 Weeks)
Contact Information
- USA: +1-678-302-0700
- India: +91-120-421-9822
- Email: info@renub.com
For more details, customization requests, or sample reports, reach out to our analysts.